Cancer Immunotherapy Drug Discovery Outsourcing Market
Cancer Immunotherapy Drug Discovery Outsourcing Market Share & Trends Analysis Report By Service type (Target Identification and Validation, Lead Screening and Characterization, Cell-based Assays, Preclinical Development, Clinical Trial Support), By Drug Type (Monoclonal Antibodies, Immunomodulators, Cancer Vaccines and Oncolytic Viral Therapy, Others), By Cancer Type (Lung Cancer, Breast Cancer, Colorectal Cancer, Melanoma, Prostate Cancer, Head & Neck Cancer, Ovarian Cancer, Pancreatic Cancer, Others) Industry Analysis Report, Regional Outlook, Growth Potential, Price Trends, Competitive Market Share & Forecast, 2025–2033

Historical Period: 2019-2024
Forecast Period: 2025-2033
Report Code : ASIPHR1011
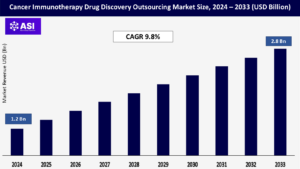
CAGR: 9.8%
Last Updated : July 15, 2025
The worldwide market for Cancer Immunotherapy Drug Discovery Outsourcing Market was valued at approximately USD 1.2 billion in 2024 and is projected to reach USD 2.8 billion by 2033, demonstrating a compound annual growth rate (CAGR) of 9.8% during the forecast period of 2025–2033.
The Cancer Immunotherapy Drug Discovery Outsourcing Market is seeing remarkable growth and it’s no surprise. As the global demand for better, more effective cancer treatments continues to rise, pharmaceutical companies are finding it increasingly challenging to handle every aspect of drug development on their own. The process is complex, time-consuming, and requires highly specialized expertise. To stay ahead, many are turning to outsourcing. By collaborating with external research partners, companies can access advanced technologies, accelerate timelines, and reduce development costs. This approach not only makes innovation more efficient but also helps bring life-saving therapies to patients faster. Industry reports tracking trends from 2025 to 2033 highlight how this shift toward outsourcing is reshaping the landscape of cancer drug discovery and it’s expected to keep growing strong in the years ahead.
Immunotherapy is gaining significant momentum in the fight against cancer, and for good reason. Treatments like immune checkpoint inhibitors, CAR-T cell therapies, and monoclonal antibodies have shown impressive results across various cancer types. Their promising efficacy has not only led to wider adoption but has also sparked a surge in the development of new therapies.
One of the key advantages of immunotherapy is its targeted nature, which in some cases results in fewer and less severe side effects compared to traditional treatments like chemotherapy. This has led to greater acceptance among both patients and healthcare providers. Moreover, immunotherapies are proving to be valuable in reducing the risk of tumor recurrence after chemotherapy, positioning them as a powerful tool for long-term cancer control.
Perhaps most importantly, they are offering new hope in cases where other treatments have failed especially for hard-to-treat or previously unresponsive cancers helping to address critical gaps in care.
Outsourcing is becoming an increasingly smart move for drug developers—and for good reason. One major advantage is cost-effectiveness. Partnering with Contract Research Organizations (CROs), especially in regions like Asia-Pacific, allows companies to significantly cut down on research and development expenses.
These partnerships offer high-quality services at a lower cost, helping organizations stretch their R&D budgets further without compromising innovation. There’s also the benefit of access to a diverse and global talent pool. By outsourcing, pharmaceutical companies can collaborate with top scientists, researchers, and clinicians from around the world. This not only brings fresh perspectives to problem-solving but also ensures that the best minds are working on developing the next generation of cancer therapies. Lastly, favorable regulatory environments in certain regions make the outsourcing process even more appealing.
In some countries, regulations around clinical trials and drug development are more streamlined and supportive, allowing companies to move through the approval process more efficiently—saving both time and money. Altogether, these factors make outsourcing not just a cost-cutting strategy, but a powerful way to accelerate progress in cancer immunotherapy.
While immunotherapy holds tremendous promise, one of the biggest hurdles remains its high cost both in development and treatment. Creating new immunotherapies is an incredibly complex and expensive process. Even with the growing trend of outsourcing to reduce costs, companies still face significant expenses tied to rigorous research, lengthy preclinical studies, and multi-phase clinical trials.
Once a therapy is approved, the price tag doesn’t drop. In fact, many immunotherapy treatments come with steep costs that can create real barriers for patients and healthcare providers alike. These high prices often raise concerns about insurance coverage and reimbursement, limiting access for many who could benefit most.
Adding to the challenge, the manufacturing of these therapies especially cutting-edge options like cell-based treatments—is complicated, resource-intensive, and hard to scale. All these factors contribute to the financial burden and can even dampen enthusiasm for investing in the next wave of innovation.
| Report Metric | Details |
|---|---|
| Segmentations | |
| By Service Type |
Target Identification and Validation Lead Screening and Characterization Cell-based Assays Preclinical Development Clinical Trial Support |
| By Drug Type |
Monoclonal Antibodies Immunomodulators Cancer Vaccines Oncolytic Viral Therapy Others |
| By Cancer Type |
Lung Cancer Breast Cancer Colorectal Cancer Melanoma Prostate Cancer Head & Neck Cancer Ovarian Cancer Pancreatic Cancer Others |
| Key Players |
|
| Geographies Covered | |
| North America |
U.S. |
| Europe |
U.K. |
| Asia Pacific |
China |
| Middle East & Africa |
Saudi Arabia |
| Latin America |
Brazil |
The Cancer Immunotherapy Drug Discovery Outsourcing Market is categorized by service type, by drug type, and by cancer type. Each segment is rapidly evolving, with different areas of the industry growing at their own pace and shifting in how much of the market they represent.
In the cancer immunotherapy drug discovery outsourcing market, companies often collaborate with specialized partners across different stages of the drug development process to streamline operations, access niche expertise, and reduce costs. One of the most critical early phases is target identification and validation.
Outsourcing this phase allows companies to harness advanced bioinformatics tools and expert assay development without needing to build this capacity in-house. The ongoing discovery of new cancer-related targets continues to fuel demand in this area. Following target validation, the focus shifts to lead screening and characterization. This phase involves screening large libraries of compounds to identify those that show promise in interacting with the target.
As the immunotherapy space continues to evolve, the complexity of preclinical models is also increasing, prompting more companies to outsource this work to experienced partners who can manage the intricate testing requirements. Finally, clinical trial support plays a vital role in bringing immunotherapy drugs to market.
When looking at outsourcing in cancer immunotherapy drug discovery, it’s helpful to consider the type of therapy being developed, as each brings its own unique set of requirements and opportunities. Monoclonal antibodies (mAbs) currently dominate this space. These lab-designed antibodies are tailored to target cancer cells or key immune checkpoints like PD-1, PD-L1, and CTLA-.
Cancer vaccines and oncolytic viral therapies represent some of the most innovative and emerging segments. Cancer vaccines are designed to prompt the immune system to recognize and attack cancer-specific antigens, while oncolytic viral therapies use genetically engineered viruses to infect and kill cancer cells triggering a broader immune response in the process. Though still niche compared to mAbs or cell therapies, both areas are seeing a surge in R&D investment, particularly with the development of personalized cancer vaccines and cutting-edge viral delivery systems.
Perhaps the most technically demanding segment is cell therapy, especially approaches like CAR-T and TCR-T, where a patient’s own immune cells are modified to seek out and destroy cancer cells. These therapies have shown groundbreaking results in certain blood cancers, driving intense interest and a rapidly growing development pipeline.
Outsourcing in cancer immunotherapy drug development is often driven by the specific types of cancers being targeted, each bringing unique challenges and opportunities. Lung cancer remains at the forefront, accounting for a significant portion of the market. Similarly, breast cancer has emerged as a key focus area, with growing attention on immunotherapy for aggressive subtypes like triple-negative breast cancer (TNBC), where traditional treatment options have often fallen short.
Melanoma holds a special place in the immunotherapy landscape as one of the earliest success stories. Meanwhile, colorectal cancer is seeing targeted interest, particularly in cases with high microsatellite instability (MSI-H), a biomarker that makes tumors more responsive to immune-based treatments.
In prostate cancer, especially in its advanced and metastatic stages, immunotherapy is being explored as a complementary option to existing therapies. These diverse efforts underscore the broad potential of immunotherapy and highlight why outsourcing remains crucial to support the complex, multi-faceted development process across cancer types.
North America, and particularly the United States, stands as the undisputed leader in the global cancer immunotherapy drug discovery outsourcing market. This dominance is not by chance it’s the result of a powerful combination of industrial strength, scientific innovation, and strategic investment. The region is home to a dense concentration of major pharmaceutical giants, pioneering biotech startups, and a well-established network of contract research organizations (CROs), creating a thriving ecosystem for drug discovery and development.
One of the region’s most compelling advantages is the scale of its investment in research and development. Both private enterprises and public institutions pour billions into cancer research, with immunotherapy being a top priority. These funds fuel a pipeline of innovation supported by cutting-edge research facilities, world-class academic institutions, and renowned cancer centers, all of which help translate scientific breakthroughs into promising therapies more rapidly.
Although the U.S. regulatory environment especially under the FDA is known for its stringency, it is also structured to encourage innovation. Accelerated approval pathways for breakthrough therapies often reduce time-to-market and incentivize outsourcing, especially in areas requiring fast, specialized development. This is especially critical in a country with a high and growing cancer burden, where the demand for novel and more effective treatments is constant
Europe stands as the second-largest player in the global cancer immunotherapy drug discovery outsourcing market, driven by a dynamic mix of scientific excellence, strategic funding, and a collaborative research culture.
Countries such as Germany, the UK, France, Switzerland, and those in the Nordic region are leading the charge, each bringing unique strengths to the table. In essence, Europe’s combination of scientific depth, collaborative ethos, and supportive regulation ensures it remains a powerhouse in cancer immunotherapy outsourcing, second only to North America.
The Asia-Pacific region is emerging as the fastest-growing market in the global cancer immunotherapy drug discovery outsourcing landscape, with countries like China, India, Japan, and South Korea at the forefront of this momentum. This surge in growth is fueled by a combination of economic, demographic, and strategic factors that are reshaping the global outsourcing map. One of the most compelling advantages Asia-Pacific offers is cost-effectiveness.
Operational and labor costs in this region are significantly lower than in North America or Europe, making it a prime destination for companies seeking to streamline their drug discovery budgets without compromising on quality. This cost advantage is particularly attractive for labor-intensive activities like preclinical research and large-scale clinical trials. The region’s vast and diverse population provides an added edge, offering an abundant pool of patients for recruitment in clinical trials—especially valuable in immunotherapy, where diverse genetic profiles and large sample sizes are crucial.
At the same time, local governments are ramping up their investments in R&D and creating environments conducive to innovation. Countries like China and India are actively supporting the growth of their biopharmaceutical sectors through tax incentives, grants, and favorable regulatory reforms. Another key driver is the emergence of a highly skilled workforce.
The Middle East and Africa region represents a nascent yet promising frontier in the cancer immunotherapy drug discovery outsourcing market. While still in the early stages of development compared to more mature regions, the groundwork is being laid for meaningful and sustained growth. One of the primary drivers of this emerging market is the rising incidence of cancer across the region.
As populations grow and lifestyles change, the burden of cancer has become more pronounced, prompting governments and healthcare providers to seek more advanced treatment solutions. Immunotherapy, with its potential to revolutionize cancer care, is gaining traction as a viable and increasingly necessary option. Governments in countries such as the UAE and Saudi Arabia are responding with significant investments aimed at modernizing their healthcare systems.
These efforts are not only designed to improve domestic care but also to position these nations as competitive players in medical tourism and regional centers for R&D. High-profile initiatives, such as the construction of cutting-edge medical cities and research parks, are clear indicators of this strategic intent.
The Cancer Immunotherapy Drug Discovery Outsourcing market was valued at USD 1.29 billion in 2024.
The Cancer Immunotherapy Drug Discovery Outsourcing market is projected to grow at a CAGR of 9.86% from 2025 to 2033.
The Monoclonal antibodies segment holds the largest market share.
The Asia-Pacific region is expected to witness the highest growth rate in Cancer Immunotherapy Drug Discovery Outsourcing market.
Major players include Covance, Inc., Horizon Discovery Group PLC, Crown Bioscience, Inc., Promega Corporation, HD Biosciences Co., Ltd., BPS Bioscience, Inc., Genscript Biotech Corporation, DiscoverX Corporation, and Explicyte.
1.1 Summary
1.2 Research methodology
2.1 Research Objectives
2.2 Market Definition
2.3 Limitations & Assumptions
2.4 Market Scope & Segmentation
2.5 Currency & Pricing Considered
3.1 Drivers
3.2 Geopolitical Impact
3.3 Human Factors
3.4 Technology Factors
4.1 Porters Five Forces Analysis
4.2 Value Chain Analysis
4.3 Average Pricing Analysis
4.4 M & A, Agreements & Collaboration Analysis
5.1 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Service Type
5.1.1 Introduction
5.1.2 Market Size & Forecast
5.2 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Drug Type
5.3 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Cancer Type
6.1 North America Cancer Immunotherapy Drug Discovery Outsourcing Market, By Country
6.1.1 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Service Type
6.1.2 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Drug Type
6.1.3 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Cancer Type
6.2 U.S.
6.2.1 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Service Type
6.2.2 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Drug Type
6.2.3 Cancer Immunotherapy Drug Discovery Outsourcing Market, By Cancer Type
6.3 Canada
7.1 U.K.
7.2 Germany
7.3 France
7.4 Spain
7.5 Italy
7.6 Russia
7.7 Nordic
7.8 Benelux
7.9 The Rest of Europe
8.1 China
8.2 South Korea
8.3 Japan
8.4 India
8.5 Australia
8.6 Taiwan
8.7 South East Asia
8.8 The Rest of Asia-Pacific
9.1 UAE
9.2 Turkey
9.3 Saudi Arabia
9.4 South Africa
9.5 Egypt
9.6 Nigeria
9.7 Rest of MEA
10.1 Brazil
10.2 Mexico
10.3 Argentina
10.4 Chile
10.5 Colombia
10.6 Rest of Latin America
11.1 Global Market Share (%) By Players
11.2 Market Ranking By Revenue for Players
11.3 Competitive Dashboard
11.4 Product Mapping