Aseptic Pharma Processing Equipment Market
Aseptic Pharma Processing Equipment Market Share & Trends Analysis Report, By Component (Processing Equipment (Dryers, Extruders, Mills, Granulators, Tablet Presses, Feeders, Fill/Finish, Mixers, Conveyors), Packaging Equipment (Inspection, Labeling, Aseptic Packaging)), By Technology (Fully-Automatic, Semi-Automatic, Single-Use Systems, Isolators, Transfer Systems),By Application (Sterile Injectables (Vials, Pre‑filled Syringes), Ophthalmic Suspensions, Lyophilized Powders, Aqueous Aerosols, Others), By End User (Pharmaceutical Manufacturers, Biotech Firms, Contract Manufacturing Organizations (CMOs), Research Laboratories)– Industry Analysis Report, Regional Outlook, Growth Potential, Price Trends, Competitive Market Share & Forecast, 2025–2033.

Historical Period: 2019-2024
Forecast Period: 2025-2033
Report Code :
CAGR: 7.50%
Last Updated : November 15, 2025
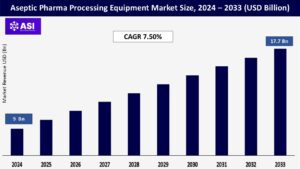
The global aseptic pharma processing equipment market size was valued at approximately USD 9 billion in 2024 and is projected to reach USD 17.7 billion by 2033, growing at a CAGR of 7.50% during the forecast period (2025–2033).
The Aseptic Pharmaceutical Processing Equipment Market involves machinery and technologies used to sterilize pharmaceutical products and maintain sterility throughout the manufacturing, filling, and packaging processes. Aseptic processing is crucial for producing sterile drugs especially biologics, injectables, vaccines, and eye drops where terminal sterilization is not feasible. This market includes equipment like cleanroom systems, isolators, sterile filling lines, autoclaves, dry heat sterilizers, filtration systems, and clean-in-place (CIP) and steam-in-place (SIP) systems. These systems are designed to eliminate contamination risks by preventing exposure of sterile products to the external environment. The primary use of this equipment is in ensuring the microbiological integrity of pharmaceutical products during critical stages of production.
The key properties of aseptic equipment include high-grade material compatibility (e.g., stainless steel, borosilicate glass), precision controls, automated monitoring, and compliance with regulatory standards such as cGMP (current Good Manufacturing Practices) and FDA or EMA guidelines. Growth in this market is driven by the rise in biologics production, demand for personalized medicines, and stringent regulations around product sterility and safety. Major players in this segment include Bosch Packaging, IMA Group, Bausch+Ströbel, Syntegon, and Getinge Group.
Modern pharmaceuticals are increasingly focused on biologics, vaccines, and injectable therapeutics, which are highly sensitive to contamination and require sterile conditions throughout production.
Unlike traditional small-molecule drugs, biologics cannot be terminally sterilized due to their delicate molecular structure. This shift has created strong demand for aseptic processing equipment to maintain sterility without compromising the product. Furthermore, the rise in personalized medicines, gene therapies, and monoclonal antibodies has boosted investments in advanced aseptic technologies, including robotics, isolators, and barrier systems.
Global regulatory authorities like the FDA (U.S.), EMA (Europe), and WHO have enforced strict cGMP (current Good Manufacturing Practice) guidelines for pharmaceutical manufacturing, particularly for sterile products. Companies must ensure zero-contamination environments, making aseptic processing essential for compliance.
Regulatory pressures have led to the modernization of production facilities with automated, closed-loop systems, reducing human intervention and enhancing product safety. This has become a significant driver, compelling pharma manufacturers to adopt state-of-the-art aseptic technologies to meet global quality standards and avoid costly recalls or penalties.
The cost barrier associated with aseptic pharmaceutical processing equipment is a major market restraint. Setting up and maintaining cleanrooms, isolators, sterile filling lines, and automated aseptic systems involves significant capital expenditure.
These technologies must adhere to stringent regulatory and validation protocols, requiring extensive documentation, qualification steps (IQ/OQ/PQ), and ongoing operational compliance. For small to mid-sized pharmaceutical manufacturers or CDMOs (Contract Development and Manufacturing Organizations), the cost and complexity of adopting these systems can be prohibitive.
In addition to high initial investment, the training of specialized staff and maintenance of sterile environments further increase operational costs. This limits the adoption of advanced aseptic processing technologies in emerging markets and among smaller players, potentially slowing overall industry growth.
| Report Metric | Details |
|---|---|
| Segmentations | |
| By Component |
Processing Equipment (Dryers, Extruders, Mills, Granulators, Tablet Presses, Feeders, Fill/Finish, Mixers, Conveyors) Packaging Equipment (Inspection, Labeling, Aseptic Packaging) |
| By Technology |
Fully-Automatic Semi-Automatic Single-Use Systems Isolators Transfer Systems |
| By Application |
Sterile Injectables (Vials, Pre‑filled Syringes) Ophthalmic Suspensions Lyophilized Powders Aqueous Aerosols Others |
| By End User |
Pharmaceutical Manufacturers Biotech Firms Contract Manufacturing Organizations (CMOs) Research Laboratories |
| Key Players |
|
| Geographies Covered | |
| North America |
U.S. |
| Europe |
U.K. |
| Asia Pacific |
China |
| Middle East & Africa |
Saudi Arabia |
| Latin America |
Brazil |
The Aseptic Pharma Processing Equipment Market is segmented by Component, Technology, Application and End User. Each factor plays a critical role in ensuring product sterility, safeguarding patient safety, and enabling the large-scale production of biologics, injectables, and sterile drug products.
The increasing regulatory focus on contamination control, coupled with innovation in automated and closed-system technologies, is accelerating the adoption of advanced aseptic processing equipment across the pharmaceutical and biotechnology industries.
Processing Equipment Includes Dryers, Extruders, Mills, Granulators, Tablet Presses, Feeders, Fill/Finish Systems, Mixers, Conveyors Dominant Segment due to its essential role in ensuring sterile drug formulation and production. Fill/Finish systems are especially critical in aseptic manufacturing, maintaining product sterility during vial/syringe filling. Mixers and granulators are key in blending active pharmaceutical ingredients (APIs) without compromising sterility. Equipment like dryers and extruders support continuous sterile manufacturing and are being upgraded for compatibility with sensitive biologics.
Packaging Equipment Includes Inspection Systems, Labeling Machines, Aseptic Packaging Units Rapidly growing segment, driven by increasing demand for ready-to-administer injectables and unit-dose formats. Aseptic packaging ensures contamination-free sealing of final products like vials and syringes. Inspection systems are essential for automated quality assurance, detecting particulate contamination and fill-volume deviations.
Fully-Automatic Systems Leading segment due to high efficiency, low contamination risk, and regulatory compliance. Favored for large-scale biologics and vaccine manufacturing, where minimizing human interaction is critical. Semi-Automatic System Ideal for mid-sized manufacturers or low-volume production.
Offers a balance between cost-effectiveness and automation but still requires manual interventions at some stages. Single-Use Systems Experiencing significant growth, particularly in biologics and personalized medicine. Reduce cleaning validation time, contamination risk, and downtime between batches.
Increasingly adopted in modular and flexible manufacturing setups. Isolators Provide a fully enclosed aseptic barrier between the product and operator. Widely adopted for high-potency drugs, toxic substances, and sterile injectables. Enhances sterility assurance and reduces contamination risks. Transfer Systems Crucial for maintaining product integrity during movement between clean zones or processes. Includes rapid transfer ports (RTPs), cleanroom pass-throughs, and automated guided vehicles (AGVs) in advanced facilities.
Sterile Injectables (Vials, Pre-filled Syringes) Largest and fastest-growing application, driven by the surge in biologics, vaccines, and monoclonal antibodies. Prefilled syringes offer convenience, accuracy, and reduced contamination risk—boosting demand for specialized aseptic filling equipment. Ophthalmic Suspensions Sensitive to microbial contamination, requiring strict aseptic conditions during filling and sealing. Growth is supported by rising incidences of ocular diseases and aging populations. Lyophilized Powders Requires specialized aseptic equipment for freeze-drying under sterile conditions.
Commonly used in biologics and vaccines to improve shelf life and stability. Aqueous Aerosols Used in inhalation therapies for respiratory conditions like asthma and COPD. Requires sterile metered-dose packaging systems to maintain drug efficacy. Others Includes parenteral nutrition, biologic suspensions, and topical sterile formulations. Segment is small but growing due to the expansion of complex drug formulations and delivery methods.
Pharmaceutical Manufacturers Primary users of aseptic processing equipment. Driven by the need for regulatory compliance, scale-up capability, and global supply chain management. Biotech Firms High growth segment, especially in monoclonal antibody, cell & gene therapy, and RNA-based therapy production. Often favor modular, flexible systems such as single-use bioreactors and automated isolators. Contract Manufacturing Organizations (CMOs) Increasingly relied upon by pharma and biotech companies for outsourced sterile manufacturing.
Invest in advanced aseptic technologies to meet diverse client requirements and ensure fast turnaround times. Research Laboratories Smaller segment but vital for clinical trial production, early-stage formulation development, and small-batch sterile trials. Require compact, flexible, and modular equipment for R&D adaptability.
North America dominates the global aseptic pharmaceutical processing equipment market, driven by its advanced pharmaceutical industry, strong regulatory framework, and widespread adoption of biologics and sterile injectables.
The United States leads in both technological innovation and demand for high-precision aseptic systems, with key players like Pfizer, Johnson & Johnson, and Moderna investing heavily in sterile manufacturing capacities—especially post-COVID-19.
The region is also home to several top aseptic equipment manufacturers, including Syntegon, SP Scientific, and GEA Group, who continuously innovate in line with FDA and cGMP standards. Canada supports market growth with an emerging biosimilar and vaccine manufacturing sector, increasingly adopting automated aseptic technologies to meet global standards.
Europe is the second-largest market, supported by a strong pharmaceutical manufacturing base in countries like Germany, Switzerland, France, Italy, and the UK. The presence of major global pharma companies, including Roche, Novartis, Sanofi, and GSK, has driven demand for sophisticated aseptic systems.
Europe’s emphasis on compliance with EMA and EU GMP guidelines pushes manufacturers to adopt advanced isolators, automated filling lines, and cleanroom technologies. Moreover, government support for innovation in sterile biologics and biosimilars, along with public-private initiatives to modernize pharmaceutical infrastructure, is accelerating growth. Environmental sustainability and energy efficiency in aseptic operations are also becoming key market differentiators in the region.
The Asia-Pacific region is the fastest-growing market for aseptic pharmaceutical processing equipment, driven by rapid pharmaceutical industrialization, expanding biopharma sectors, and government incentives. China, India, Japan, and South Korea are the major contributors.
China is making massive investments in domestic vaccine and biologics production, prompting demand for aseptic isolators, single-use systems, and cleanroom setups. India, a global hub for generic and contract drug manufacturing, is increasingly adopting aseptic technologies to meet FDA and EU inspection standards.
Japan and South Korea, with strong R&D capabilities, are leveraging cutting-edge automation and robotics in sterile drug production. The region’s growing middle-class population and demand for injectable therapeutics are further supporting long-term market expansion.
Latin America shows moderate but steady growth, with Brazil, Mexico, and Argentina leading the regional market. The pharmaceutical sector is expanding due to rising healthcare needs, the push for local vaccine production, and increased outsourcing by multinational pharma firms.
However, adoption of advanced aseptic equipment is still limited by budget constraints, regulatory complexity, and lack of infrastructure. Despite these challenges, regional governments are investing in pharmaceutical modernization programs and forming partnerships with international equipment suppliers to improve production standards, especially for sterile injectables and biosimilars.
The Middle East & Africa region represents a nascent but emerging market for aseptic pharmaceutical processing equipment. Countries like Saudi Arabia, the UAE, Egypt, and South Africa are taking steps to develop local pharmaceutical manufacturing capabilities, particularly in response to health security concerns and rising chronic disease burdens.
The Gulf Cooperation Council (GCC) nations are investing in state-of-the-art sterile production plants, with support from foreign pharma and biotech investors. However, much of sub-Saharan Africa still lacks the technological and regulatory infrastructure needed for advanced aseptic processing. Growth in this region is expected to be driven by government health initiatives, international collaborations, and increased demand for vaccines and parenteral drugs.
The aseptic pharma processing equipment market was valued at USD 9 billion in 2024.
The aseptic pharma processing equipment market is projected to grow at a CAGR of 7.50% from 2025 to 2033.
The Processing Equipment hold the largest aseptic pharma processing equipment market share.
The North America region is expected to witness the highest growth rate.
The Major players include Syntegon Technology GmbH, IMA Group and Bausch+Ströbel Maschinenfabrik Ilshofen GmbH + Co. KG.
1.1 Summary
1.2 Research methodology
2.1 Research Objectives
2.2 Market Definition
2.3 Limitations & Assumptions
2.4 Market Scope & Segmentation
2.5 Currency & Pricing Considered
3.1 Drivers
3.2 Geopolitical Impact
3.3 Human Factors
3.4 Technology Factors
4.1 Porters Five Forces Analysis
4.2 Value Chain Analysis
4.3 Average Pricing Analysis
4.4 M & A, Agreements & Collaboration Analysis
5.1 Aseptic Pharma Processing Equipment Market, By Component
5.1.1 Introduction
5.1.2 Market Size & Forecast
5.2 Aseptic Pharma Processing Equipment Market, By Technology
5.3 Aseptic Pharma Processing Equipment Market, By Application
5.4 Aseptic Pharma Processing Equipment Market, By End User
6.1 North America Aseptic Pharma Processing Equipment Market , By Country
6.1.1 Aseptic Pharma Processing Equipment Market, By Component
6.1.2 Aseptic Pharma Processing Equipment Market, By Technology
6.1.3 Aseptic Pharma Processing Equipment Market, By Application
6.1.4 Aseptic Pharma Processing Equipment Market, By End User
6.2 U.S.
6.2.1 Aseptic Pharma Processing Equipment Market, By Component
6.2.2 Aseptic Pharma Processing Equipment Market, By Technology
6.2.3 Aseptic Pharma Processing Equipment Market, By Application
6.2.4 Aseptic Pharma Processing Equipment Market, By End User
6.3 Canada
7.1 U.K.
7.2 Germany
7.3 France
7.4 Spain
7.5 Italy
7.6 Russia
7.7 Nordic
7.8 Benelux
7.9 The Rest of Europe
8.1 China
8.2 South Korea
8.3 Japan
8.4 India
8.5 Australia
8.6 Taiwan
8.7 South East Asia
8.8 The Rest of Asia-Pacific
9.1 UAE
9.2 Turkey
9.3 Saudi Arabia
9.4 South Africa
9.5 Egypt
9.6 Nigeria
9.7 Rest of MEA
10.1 Brazil
10.2 Mexico
10.3 Argentina
10.4 Chile
10.5 Colombia
10.6 Rest of Latin America
11.1 Global Market Share (%) By Players
11.2 Market Ranking By Revenue for Players
11.3 Competitive Dashboard
11.4 Product Mapping