Biobanking Market
Biobanking Market size, share, & trends analysis report By Sample Type (Human Tissues and Organs ,Blood and Blood Components (Plasma, Serum, Whole Blood) Cell Lines, Nucleic Acids (DNA, RNA), Others ) By Application (Regenerative Medicine ,Life Science Research ,Clinical Diagnostics, Drug Discovery & Development, Personalized Medicine), By End User(Academic & Research Institutions, Pharmaceutical & Biotechnology Companies, Hospitals &Clinics, Contract Research Organizations (CROs),Government & Public Health Organizations) Industry Analysis Report, regional outlook growth potential, price trends, competitive market share and & forecast period 2025-2033.

Historical Period: 2019-2024
Forecast Period: 2025-2033
Report Code :
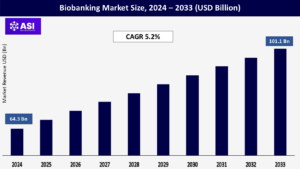
CAGR: 5.2%
Last Updated : February 25, 2026
The global biobanking market was valued at approximately USD 64.3 billion in 2024 and is projected to reach USD 101.1 billion by 2033, growing at a CAGR of 5.2% during the forecast period (2025–2033).
Biobanking involves the process of collecting, storing, and managing biological samples such as blood, tissues, DNA, and cells, along with associated health data, for use in research and clinical applications. To retain their integrity for subsequent analysis in genomics, drug development, disease processes, and individualized medicine studies, biospecimens are kept under closely regulated conditions. Through the supply of quality, well-annotated specimens that support targeted medicine development, diagnosis of early illness, and precision diagnostics, biobanks contribute to speeding up medical research. The increasing burden of chronic diseases, the increased demand for personalized medicine, and advances in regenerative therapies and genomics research are propelling the industry.
The demand for biobanking facilities is especially being driven by rising investments in genomic and biomedical research. Large-scale genomic mapping and population health research are being funded by governments, academic institutions, and business organizations; these projects all depend on well-preserved, high-quality biosamples and related metadata. Because they provide standardized, long-term storage of biospecimens needed to investigate genetic predispositions, disease processes, and treatment responses, biobanks are essential to supporting such efforts. One good example is the UK 100,000 Genomes Project, which used samples from patients who were receiving treatment via the National Health Service in order to enhance understanding about rare diseases and cancer. The project has since been renamed Genomics England and is still pushing innovation in precision medicine.
In the same vein, China’s National Gene Bank and the United States’ NIH All of Us Research Program are collecting millions of samples in order to drive personalized health care. Such efforts require strong biobanking infrastructure with cutting-edge informatics and ethical data-sharing policies. With genomic sequencing becoming more affordable and accessible, and gene-based therapy studies picking up speed, biobanks are emerging as the pillars of translational medicine and next-generation diagnostics.
Increased global interest in personalized medicine is one of the main drivers fueling biobanking growth. Personalized medicine aims to tailor treatment plans based on individuals’ genetic makeup, lifestyle, and environmental exposures. Biobanks provide the underlying support infrastructure for this healthcare revolution by sampling, storing, and distributing high-quality biological materials such as blood, tissue, and DNA together with detailed phenotypic and clinical information.
These samples are invaluable in genomic studies, biomarker discovery, and drug development for targeted patient subgroups. In 2023, more than 40% of all new drug approvals by the U.S. FDA were categorized as personalized medicines, reports the Personalized Medicine Coalition. A great example is the UK Biobank, with over 500,000 participants who have contributed data to enable disease prevention and precision diagnosis.
Pharma companies and research institutions are increasingly leveraging such biobank collections to develop targeted medicines, particularly in oncology, neurology, and genetic rare diseases. As health systems globally adopt precision medicine strategies, demand for large, heterogeneous, and well-annotated biobank collections will significantly rise and drive market growth.
While biobanking plays a pivotal role in advancing personalized medicine and biomedical research, ethical and legal complexities pose a significant restraint on the market’s growth. One of the central concerns revolves around the collection, storage, and secondary use of human biological samples, which often raise questions related to informed consent, privacy, and data protection. In many cases, donors are asked to give broad or unspecified consent for future research, which may include commercial use or studies not initially envisioned a practice that has been criticized for being ethically ambiguous.
The situation is further complicated by variability in international regulations. For example, under the European Union’s General Data Protection Regulation (GDPR), stringent rules govern how personal data, including genetic information, can be processed and shared. Non-compliance can result in severe penalties, which makes multinational research collaborations more difficult and expensive. A 2022 study published in Bio preservation and Biobanking highlighted that over 60% of surveyed biobanks faced regulatory challenges that delayed sample sharing or research projects.
| Report Metric | Details |
|---|---|
| Segmentations | |
| By Sample Type |
Human Tissues and Organs Blood and Blood Components (Plasma, Serum, Whole Blood) Cell Lines Nucleic Acids (DNA, RNA) Others (Urine, Saliva, Microbial Samples) . |
| By Application |
Regenerative Medicine Life Science Research Clinical Diagnostics Drug Discovery & Development Personalized Medicine |
| By End User |
Academic & Research Institutions Pharmaceutical & Biotechnology Companies Hospitals & Clinics Contract Research Organizations (CROs) Government & Public Health Organizations |
| Key Players |
|
| Geographies Covered | |
| North America |
U.S. |
| Europe |
U.K. |
| Asia Pacific |
China |
| Middle East & Africa |
Saudi Arabia |
| Latin America |
Brazil |
Blood and Blood Products (Plasma, Serum, Whole Blood) lead the biobanking market, capturing the greatest share in 2024. Their extensive application in clinical diagnostics, genomics, proteomics, and biomarker discovery fuels demand.
Blood samples are easy to obtain, highly informative, and are crucial in almost all fields of biomedical investigation and drug development, making them a staple of biobanking activities. Human Tissues and Organs are a major and expanding business area driven by the rising interest in precision medicine and tissue-specific research. These tissues are crucial for research on cancer, regenerative medicine, and histopathology, offering detailed information about mechanisms of disease at the cellular level.
Cell Lines are acquiring momentum as they play a key role in drug screening, toxicity testing, and targeted or personalized medicine. Immortalized human and animal cell lines provide scalable, reproducible biological material for high-throughput assays, rendering them worth their salt for pharmaceutical companies and research institutions.
Nucleic Acids (DNA, RNA) represent an essential component as genetic and epigenetic research increases in demand. As next-generation sequencing (NGS) technologies and genome-wide association studies (GWAS) continue to advance, DNA/RNA storage and analysis have become the mainstay of contemporary biobanking.
Life Science Research has the highest market share in 2024, driven by rising academic and commercial need for high-quality bio specimens to aid genomic, proteomic, and metabolomic research. Biobanks form the backbone of basic and translational science, facilitating reproducible scientific advancement.
Drug Discovery & Development is a fast-emerging segment as pharma and biotech companies increasingly depend on biobanking samples to validate drug targets, identify biomarkers, and conduct preclinical testing. Biobanks reduce the length of drug pipelines by providing access to well-characterized biological material.
Clinical Diagnostics is poised to experience steady expansion, fueled by incorporation of biobank-derived data into diagnostic assays. Stored bio specimens facilitate the growth of molecular diagnostics, companion diagnostics, and early disease detection approaches. Regenerative Medicine is a fast-growing application segment with evolving developments in stem cell therapy and tissue engineering. Biobanking stem cells and tissues are key drivers in cell-based therapies and transplantation.
Personalized Medicine is rapidly growing, with an increasing need for customized treatment regimens based on genetic information. Biobanks make it possible to link genotypic and phenotypic information, refining clinical decision-making and individualized interventions.
Academic & Research Institutions are the predominant end-users with the highest market share in 2024. They are on the leading edge of biomedical discovery, leveraging biobanking specimens to advance research in a variety of fields, from genomics to public health. Pharmaceutical & Biotechnology Companies are also expanding their use of biobanking for R&D, biomarker qualification, and clinical trials. The need for quality-assured, annotated bio specimens to aid precision drug development is driving this segment.
Hospitals & Clinics are instrumental in biobanking, especially in the acquisition and storage of patient samples associated with electronic health records. Their participation is fundamental to disease-specific biobanks and clinical outcome studies, connecting patient care and research. Contract Research Organizations (CROs) are expanding their presence within the biobanking space as they facilitate sample management, storage, and downstream analysis for pharmaceutical clients. Their involvement in outsourced biobanking services keeps increasing as pharma focuses on reducing costs and simplifying operations.
Government & Public Health Organizations are prominent players in large-scale population biobanking initiatives. National and regional biobanks, usually funded by public agencies, facilitate epidemiological studies and disease surveillance programs, particularly during pandemics and prevention of chronic disease.
North America is leading the global biobanking market right now with an estimated 38.5% share in 2024. The leading factor is strong government support, a well-developed research base, and high precision medicine and genomic study adoption. The United States leads the region due to significant initiatives such as the NIH’s “All of Us” Research Program, which aims to collect bio specimens from over one million people for personalized healthcare studies.
Furthermore, the presence of major pharmaceutical companies, biobanks like the Mayo Clinic Biobank and NCI’s Cancer Human Biobank (caHUB), and favorable regulatory frameworks support market growth. Strong awareness regarding tissue donation and excellent partnerships among academic institutions and commercial biobanks significantly support the U.S. market.
Europe is second in the world with the largest share in the biobanking industry, and nations such as the UK, Germany, Sweden, and the Netherlands make substantial contributions. Long-standing biobank infrastructures such as the UK Biobank, Estonian Genome Centre, and Biobank Graz in Austria are enjoyed by the region.
In 2024, Horizon Europe and BBMRI-ERIC (Biobanking and Bio Molecular resources Research Infrastructure – European Research Infrastructure Consortium) initiatives persist in supporting data harmonization, ethical requirements, and biobank collaboration across borders. Population aging, the rise in biomedical research spending, and the pervasiveness of chronic disease burden are the primary drivers of the growth of the market in the region.
The biobanking market in the Asia-Pacific region is anticipated to grow at the highest rate during the forecast period, with a CAGR of 8.2% from 2024 to 2030. This growth is driven by increasing genomics research, healthcare investments, and favorable government policies in the emerging markets.
China has hugely invested in biobanking infrastructure through the National GeneBank, whereas Japan and South Korea have initiated population-scale genomics projects with EHRs. India’s GenomeIndia program, to sequence 10,000 individuals, is yet another example of increasing regional involvement. Growth in disease surveillance, regenerative medicine, and pharma R&D is also driving biobanking growth in the region of Asia.
Latin America and Middle East & Africa (MEA) markets are new biobanking markets, which are witnessing moderate but consistent growth. In Latin America, there is growing investment in genomic medicine and public health surveillance in countries such as Brazil and Mexico. An example of public sector engagement in sample collection for cancer research exists in the form of Brazil’s National Cancer Institute (INCA) biobank.
Within the MEA region, South Africa takes the lead with established biobanks like the National Health Laboratory Service (NHLS) Biobank, and other countries are increasingly realizing the function of biobanks in managing infectious disease and promoting local R&D. Yet, factors such as access to finance, infrastructure inequalities, and regulatory issues still limit accelerated growth within both regions.
The biobanking market was valued at USD 64.3 billion in 2024.
The biobanking market is projected to grow at a CAGR of 5.2 % from 2025 to 2033.
Human tissue biobanking hold the largest market share.
The Asia-Pacific region is expected to witness the highest growth rate.
Major players include – Thermo Fisher Scientific Inc, Azenta Life Sciences (formerly Brooks Life Sciences), Qiagen N.V., Tecan Group Ltd, Hamilton Company
1.1 Summary
1.2 Research methodology
2.1 Research Objectives
2.2 Market Definition
2.3 Limitations & Assumptions
2.4 Market Scope & Segmentation
2.5 Currency & Pricing Considered
3.1 Drivers
3.2 Geopolitical Impact
3.3 Human Factors
3.4 Technology Factors
4.1 Porters Five Forces Analysis
4.2 Value Chain Analysis
4.3 Average Pricing Analysis
4.4 M & A, Agreements & Collaboration Analysis
5.1 Biobanking Market, By Sample Type
5.1.1 Introduction
5.1.2 Market Size & Forecast
5.2 Biobanking Market, By Application
5.3 Biobanking Market, By End User
6.1 Biobanking Market, By Sample Type
6.1.1 Biobanking Market, By Sample Type
6.1.2 Biobanking Market, By Application
6.1.3 Biobanking Market, By End User
6.2 U.S.
6.2.1 Biobanking Market, By Sample Type
6.2.2 Biobanking Market, By Application
6.2.3 Biobanking Market, By End User
6.3 Canada
7.1 U.K.
7.2 Germany
7.3 France
7.4 Spain
7.5 Italy
7.6 Russia
7.7 Nordic
7.8 Benelux
7.9 The Rest of Europe
8.1 China
8.2 South Korea
8.3 Japan
8.4 India
8.5 Australia
8.6 Taiwan
8.7 South East Asia
8.8 The Rest of Asia-Pacific
9.1 UAE
9.2 Turkey
9.3 Saudi Arabia
9.4 South Africa
9.5 Egypt
9.6 Nigeria
9.7 Rest of MEA
10.1 Brazil
10.2 Mexico
10.3 Argentina
10.4 Chile
10.5 Colombia
10.6 Rest of Latin America
11.1 Global Market Share (%) By Players
11.2 Market Ranking By Revenue for Players
11.3 Competitive Dashboard
11.4 Product Mapping