Europe Cardiac Resynchronization Therapy (CRT) Market
Europe Cardiac Resynchronization Therapy (CRT) Market Share & Trends Analysis Report, By Product Type ( CRT‑Defibrillator (CRT‑D), CRT‑Pacemaker (CRT‑P)), By End‑User (Hospitals, Cardiac Centers, Other Healthcare Facilities), By Patient Type / Pacing Modality (Bi‑Ventricular Pacing, Multi‑Site Pacing, Adaptive Pacing)– Industry Analysis Report, Regional Outlook, Growth Potential, Price Trends, Competitive Market Share & Forecast, 2025–2033.

Historical Period: 2019-2024
Forecast Period: 2025-2033
Report Code :
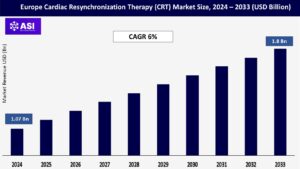
CAGR: 6.00%
Last Updated : December 16, 2025
The global Europe Cardiac Resynchronization Therapy (CRT) Market size was valued at approximately USD 1.07 billion in 2024 and is projected to reach USD 1.8 billion by 2033, growing at a CAGR of 6.00% during the forecast period (2025–2033).
In the European cardiac resynchronization therapy (CRT) market, manufacturers supply implantable devices CRT pacemakers (CRT-P) and CRT defibrillators (CRT-D) designed to correct the electrical dyssynchrony that underlies many cases of chronic heart failure.
Each system houses a pulse generator, powered by a lithium-silver-vanadium battery that can last five to ten years, and three pacing leads: one to the right atrium, one to the right ventricle, and a third fed through the coronary sinus to stimulate the left ventricular free wall.
By delivering precisely timed impulses, the device resynchronizes ventricular contractions, boosting cardiac output, easing symptoms such as dyspnea and fatigue, and reducing rehospitalization rates; CRT-D models add high-energy shocks and anti-tachycardia pacing to avert sudden cardiac death.
Contemporary European offerings emphasize MRI conditional labeling, Bluetooth-enabled remote monitoring portals compliant with GDPR, adaptive algorithms that fine-tune atrioventricular and interventricular delays, and smaller hermetically sealed titanium housings that improve patient comfort. Demand is driven by the region’s aging population, the prevalence of bundle-branch block, and reimbursement frameworks that favor therapies shown to enhance quality of life and cut long-term healthcare expenditure.
One of the strongest drivers of the cardiac resynchronization therapy market in Europe is the increasing incidence of heart failure and associated cardiovascular conditions, particularly among the aging population. Heart failure affects millions across Europe, and a significant subset of these patients exhibit electrical conduction delays especially left bundle branch block that make them eligible for CRT.
The growing burden of chronic conditions such as hypertension, diabetes, and coronary artery disease has further contributed to the rise in heart failure cases. As CRT is clinically proven to improve symptoms, reduce hospitalizations, and extend life expectancy in selected patients, its adoption has become a cornerstone of advanced heart failure management in both primary and tertiary care settings. National healthcare systems across Europe are increasingly recognizing CRT as a cost-effective intervention, supporting its inclusion in clinical guidelines and reimbursement schemes.
Technological innovation is another key driver propelling the CRT market in Europe. Modern CRT systems have evolved to become smaller, smarter, and more patient-friendly. Features such as adaptive pacing algorithms, MRI compatibility, extended battery life, and remote patient monitoring via telehealth platforms have significantly improved device performance and patient outcomes.
For instance, remote monitoring enables early detection of arrhythmias or device malfunctions, allowing timely clinical intervention and reducing the need for in-person follow-ups—an advantage particularly valued in rural or underserved regions.
In addition, the integration of artificial intelligence and predictive analytics into device software allows for personalized therapy adjustments. These innovations, supported by robust regulatory frameworks such as CE marking and EMA guidelines, have boosted physician confidence and patient acceptance, driving wider adoption of CRT therapy across Europe.
In Europe, one of the major restraints for the CRT market is the complex and evolving regulatory landscape, particularly following the implementation of the EU Medical Device Regulation (MDR). The MDR imposes stricter requirements for clinical evaluation, technical documentation, and post-market surveillance, increasing the time and cost for CRT devices to gain CE certification.
This can delay the introduction of innovative products into the European market, especially for smaller or newer manufacturers. Additionally, reimbursement policies vary widely across EU member states, and some public healthcare systems have rigid cost-benefit assessments that can restrict or delay access to CRT—especially for CRT-D devices, which are more expensive than CRT-P systems.
In certain countries, budget constraints or limited awareness among general practitioners may lead to underutilization of CRT therapy, even in eligible patients. These regulatory and reimbursement challenges can hinder market penetration and limit growth, despite the proven clinical benefits of CRT.
| Report Metric | Details |
|---|---|
| By Product Type |
CRT‑Defibrillator (CRT‑D) CRT‑Pacemaker (CRT‑P) |
| By End-User |
Hospitals Cardiac Centers Other Healthcare Facilities |
| By Patient Type / Pacing Modality |
Bi‑Ventricular Pacing Multi‑Site Pacing Adaptive Pacing |
| Key Players |
|
| Europe |
U.K. |
The Europe Cardiac Resynchronization Therapy (CRT) Market is segmented by Product Type, End User and Patient Type. Each driver, including the rising burden of heart failure and continuous innovation in device technology, plays a critical role in expanding access to life-saving cardiac therapies, improving patient quality of life, and supporting the evolution of personalized, evidence-based treatment strategies.
The market is bifurcated into CRT-Defibrillators (CRT-D) and CRT-Pacemakers (CRT-P). CRT-D devices dominate the market due to their dual functionality—providing both cardiac resynchronization and protection against sudden cardiac arrest through defibrillation.
These devices are particularly preferred for high-risk heart failure patients with a history of ventricular arrhythmias. Despite being more expensive, CRT-D units offer superior clinical outcomes in patients with reduced ejection fraction and are widely used in tertiary care centers across countries like Germany, France, and the UK.
On the other hand, CRT-P devices, which are limited to pacing therapy without defibrillation, are more cost-effective and are often prescribed for patients who do not require defibrillation support. These are commonly used in elderly populations or patients with lower arrhythmic risk, contributing to steady growth in markets where cost-sensitive healthcare decisions are prevalent, such as in Southern or Eastern Europe.
Hospitals form the largest segment, driven by the availability of cardiac electrophysiology labs, trained interventional cardiologists, and reimbursement support for device-based heart failure therapies. These institutions typically handle complex CRT implantations and follow-up care. Cardiac centers, including standalone heart institutes and specialty cardiology departments, are also major contributors to the market, particularly in Western Europe where infrastructure is more advanced.
These centers often conduct high volumes of CRT procedures and participate in clinical research and remote device monitoring programs. The other healthcare facilities segment—encompassing ambulatory surgical centers, outpatient clinics, and rehabilitation facilities—is growing steadily as follow-up management, device monitoring, and post-implantation therapy shift toward more decentralized care models, enabled by telehealth and remote diagnostics.
The CRT market is segmented into Bi-Ventricular Pacing, Multi-Site Pacing, and Adaptive Pacing. Bi-Ventricular Pacing is the most established and widely used modality, involving synchronized stimulation of the right and left ventricles to improve cardiac output and coordination.
It remains the standard of care for the majority of CRT candidates. Multi-Site Pacing, which delivers stimulation to more than one site within the same ventricle or to multiple left ventricular areas, is gaining attention for its potential to improve outcomes in patients who are non-responders to conventional CRT.
Although still under evaluation in many clinical trials, its adoption is expected to grow in high-end cardiac centers. Adaptive Pacing represents the cutting-edge segment, where intelligent algorithms dynamically adjust pacing intervals based on the patient’s real-time physiological needs.
These advanced systems aim to personalize therapy and minimize unnecessary pacing, which can improve battery life and enhance long-term outcomes. While still emerging, adaptive pacing is attracting interest in technologically advanced healthcare systems in Northern and Western Europe.
While the primary focus is on Europe, it’s important to compare the global context. North America, particularly the U.S., leads the CRT market globally due to high awareness of advanced cardiac therapies, favorable reimbursement policies, and a large patient population suffering from heart failure and arrhythmias.
The presence of leading device manufacturers and widespread use of implantable cardiac devices ensures rapid adoption of CRT technologies, including advanced CRT-D systems with remote monitoring capabilities.
Europe is a major hub for cardiac resynchronization therapy, with countries such as Germany, the UK, France, and Italy at the forefront of both clinical research and device implantation. Strong regulatory frameworks (CE marking and MDR compliance), high healthcare spending, and a growing elderly population support the growth of CRT across the continent.
Western Europe leads in adopting CRT-D and adaptive pacing modalities, while Eastern Europe, though expanding, may still lag in terms of device availability and reimbursement support. European healthcare systems increasingly favor evidence-based, cost-effective therapies, further promoting CRT adoption in eligible patients with moderate to severe heart failure.
The Asia-Pacific region is seeing growing interest in CRT as the prevalence of cardiovascular disease continues to rise in densely populated nations like China and India. However, adoption remains lower than in Europe due to higher costs, limited specialist availability, and less developed cardiac care infrastructure.
In Japan and South Korea, where healthcare systems are more advanced, CRT implantation rates are rising steadily. The increasing number of cardiac centers and growing awareness among clinicians about the benefits of CRT are expected to boost market penetration over the next decade.
In Latin America, CRT adoption is gradually increasing, particularly in Brazil, Argentina, and Mexico, where tertiary care hospitals are beginning to incorporate advanced cardiac pacing technologies. However, inconsistent reimbursement frameworks and economic constraints limit accessibility to high-cost devices like CRT-D. Public health programs often prioritize essential care, leading to selective use of CRT primarily in urban and private healthcare facilities.
The CRT market in the Middle East & Africa is still in its early stages of development. High-income nations such as the UAE, Qatar, and Saudi Arabia are adopting CRT at a growing pace due to improved healthcare investments and partnerships with Western device manufacturers.
In contrast, large parts of Africa face infrastructural challenges, limited specialist availability, and affordability issues, which hinder the broader use of implantable cardiac devices. Nonetheless, with international aid and growing cardiovascular awareness, the region shows long-term potential.
The Europe cardiac resynchronization therapy market was valued at USD 1.07 billion in 2024.
The Europe cardiac resynchronization therapy market is projected to grow at a CAGR of 6% from 2025 to 2033.
The CRT‑Defibrillator (CRT‑D) hold the largest Europe cardiac resynchronization therapy market share.
The Europe region is expected to witness the highest growth rate.
Major players include Medtronic plc, Abbott Laboratories and Boston Scientific Corporation
1.1 Summary
1.2 Research methodology
2.1 Research Objectives
2.2 Market Definition
2.3 Limitations & Assumptions
2.4 Market Scope & Segmentation
2.5 Currency & Pricing Considered
3.1 Drivers
3.2 Geopolitical Impact
3.3 Human Factors
3.4 Technology Factors
4.1 Porters Five Forces Analysis
4.2 Value Chain Analysis
4.3 Average Pricing Analysis
4.4 M & A, Agreements & Collaboration Analysis
5.1 Europe Cardiac Resynchronization Therapy (CRT) Market, By Product Type
5.1.1 Introduction
5.1.2 Market Size & Forecast
5.2 Europe Cardiac Resynchronization Therapy (CRT) Market, By End-User
5.3 Europe Cardiac Resynchronization Therapy (CRT) Market, By Patient Type / Pacing Modality
6.1 U.K.
6.2 Germany
6.3 France
6.4 Spain
6.5 Italy
6.6 Russia
6.7 Nordic
6.8 Benelux
6.9 The Rest of Europe
7.1 Global Market Share (%) By Players
7.2 Market Ranking By Revenue for Players
7.3 Competitive Dashboard
7.4 Product Mapping