Wound Care Biologics Market
Wound Care Biologics Market Share and Trend Analysis, By Technology (Growth Factors, Skin Substitutes (Allografts/Xenografts), Biomatrices, Fusion Technologies), By Application (Chronic Wounds, Acute Wounds, Surgical Wounds, Burns), By End User (Hospitals, Specialty Clinics, Home Healthcare) Industry Analysis Report, Regional Outlook, Growth Potential, Price Trends, Competitive Market Share & Forecast, 2026–2033

Historical Period: 2019-2024
Forecast Period: 2025-2033
Report Code :
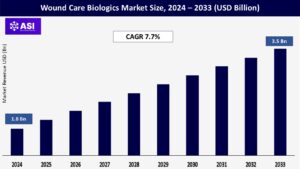
CAGR: 7.7%
Last Updated : March 31, 2026
The global Wound Care Biologics Market size was valued at USD 1.8 billion in 2024 and is projected to reach USD 3.5 billion by 2033, expanding at a compound annual growth rate CAGR of 7.7 % during the forecast period (2025 – 2033).
Wound care biologics are a revolutionary method for healing complicated wounds by utilizing biological-derived materials to activate the body’s inherent regenerative processes. In contrast to conventional techniques, these cutting-edge therapies – growth factors, cellular matrices, and skin substitutes such as placental membranes and collagen scaffolds – actively facilitate tissue healing in difficult acute and chronic wounds.
They’re especially crucial for diabetic foot ulcers, venous leg ulcers, critical burns, and non-healing surgical wounds, where standard treatments fail. By establishing a bioactive environment, biologics decrease infection risk, reduce scarring, and speed up recovery periods.
Their clinical adoption is growing at a rapid pace throughout hospitals, specialty wound centers, and home care due to the desperate need to combat growing global burdens such as diabetes, aging, and complicated trauma.
Clinicians see much improved results with biologics in treating resistant wounds, although cost continues to be an obstacle. As regenerative medicine continues to advance, these solutions provide a shift toward personalized, effective wound management that enhances long-term patient outcomes while reducing healthcare system expense.
The global epidemic of diabetes is inundating clinics with nonhealing wounds that resist even the best efforts of standard bandages. Diabetic foot ulcers alone ensnare millions in vicious cycles of pain and infection, where those very bandages may as well be wallpaper.
Biologics shatter this nightmare by providing living tissue builders – bioengineered grafts and growth factor potions that rebuild devastated skin from the inside out. They don’t simply mask damage; they mobilize the body’s repair teams to regrow nerves and blood vessels.
Physicians today are witnessing “hopeless” ulcers heal in weeks instead of lingering for years, restoring mobility to patients who’d given up wheelchairs. With exploding aging populations (more than 20% soon to be seniors), paper-thin skin makes minor scrapes become life-altering wounds.
Nurses see biologics bringing patients back from the brink of amputation every day. Health systems now count on these treatments not only to cure, but to reduce months-long hospital stays.
When a $2,000 graft that saves a $100,000 amputation – in addition to prevents lifetime disability expenses and home care burden – even budget directors sit up and take notice. This is not medicine – it’s rescue missions for lives hijacked by chronic disease, back to work and the playground for breadwinners and grandparents alike.
Regenerative medicine is transforming wound care into a real-world sci-fi revolution. Picture rolling out a “living bandage” – 3D-printed with your own cells, teeming with growth signals that navigate tissue like a biological GPS. Products such as amniotic membranes act as nature’s pharmacy, releasing enzymes that suffocate inflammation while fighting bacteria.
Small labs now cultivate grafts in bioreactors whirring 24/7, tailoring thickness for burns compared to bedsores. Startups sew micro-sensors into these grafts that text physicians: “Infection cooking – send reinforcements.”
Regulators scramble to speed up breakthroughs – such as a new spray-on stem cell foam that treated radiation burns in a matter of days. Venture capital floods labs because real-world wins go viral: the factory worker who regrew fingertip skin after a press accident, or the veteran who tossed his crutches after nerve-weaving matrices revived his shattered leg.
You’re seeing garage-born startups outpace Big Pharma, slashing production costs while boosting efficacy. What was once experimental now halts disabilities before they start. Clinics can’t get these miracles on the shelf quickly enough – a rural hospital told of grafts taking off the shelves twice as rapidly as antibiotics. Next-generation bioprinters already print tissue at the bedside, with a promise of on-demand skin in this decade.
The harsh reality of biologics strikes most painfully where it hurts the most: the wallet. These breakthrough therapies have price tags that dwarf standard wound care—conceivable paying more per application than one month’s rent, as regular dressings cost pocket money.
Insurance gatekeepers in emerging nations habitually reject them as “luxuries,” not saviors. Imagine a diabetic patient observing their ulcer deepen while approval committees argue about cost thresholds. In America, Medicare’s reimbursement maze swamps clinics in paperwork, keeping care pending until wounds become necrotic.
Europe’s strict cost-effectiveness tests immobilize innovation at the border. Small clinics? They must make heart-wrenching choices: buy one dose of a biologic or purchase material for a dozen patients. And in poor areas, biologics may as well be unicorns published in journals but never in neighborhood hospitals.
Physicians understand the heartless arithmetic: one top-of-the-line graft or nourish an entire ward for weeks. Until the payment policies fill this gulf between lab advances and bedside availability, biologics will continue to be an illusion for those hemorrhaging in waiting rooms. It’s not economics, it’s triage by spreadsheet.
| Report Metric | Details |
|---|---|
| Segmentations | |
| By Technology |
Growth Factors Skin Substitutes (Allografts/Xenografts) Biomatrices Fusion Technologies |
| By Application |
Chronic Wounds Acute Wounds Surgical Wounds Burns |
| By End Use |
Hospitals Specialty Clinics Home Healthcare |
| Key Players |
|
| Geographies Covered | |
| North America |
U.S. |
| Europe |
U.K. |
| Asia Pacific |
China |
| Middle East & Africa |
Saudi Arabia |
| Latin America |
Brazil |
Growth factors are at the center of biologic wound healing, serving as molecular messengers that yell “repair here!” to sleeping cells. Consider them as maestros leading the repair symphony of the body. PDGF mobilizes fibroblasts to restore collagen roads, while VEGF mobilizes new vessels as a kind of rescue teams.
They’re front-line fighters against resistant ulcers, speeding closure where simple dressings won’t. Skin substitutes are where architecture meets biology. Allografts, engineered from donated human tissue such as amniotic membranes, provide “off-the-shelf” healing—stuffed with antimicrobial protection and growth factors into a biological Band-Aid.
Xenografts, usually porcine collagen matrices, support injured tissue to allow native cells to repopulate. Their magic is adaptability: molding to irregular burns or padding bedsores while stemming infections. Biomatrices operate behind the scenes as regenerative blueprints.
Hyaluronic acid gels, for instance, provide moisture-laden hideaways where stem cells double and interweave new tissue. Breakthroughs such as freeze-dried placental powders now have extended shelf lives, allowing rural clinics to carry these game-changers. Combined, these drugs constitute a healing triad—signaling, shielding, and scaffolding—to convert wounds into well-being.
Chronic wounds such as diabetic ulcers are low-level wars of attrition in which gauze and antibiotics lose. Biologics engage as special forces: growth factors ambush inflamed tissue, resuming cellular repair while biomatrices remove infection. Recalcitrant pressure ulcers yield to paired offloading and collagen scaffolds enlisting regenerative reinforcements.
Venous leg ulcers yield to human amniotic grafts that serve as inflammation sponges and structural scaffolds. Acute wounds require frontline triage. Third-degree burns are addressed by spray-on skin substitutes putting out tissue fires and re-growing nerves.
Trauma wounds—crushed limbs, degloving injuries—re-grow through 3D-printed matrices that direct the regrowth like biological GPS. Surgical incisions now benefit from “healing insurance”: placental powder pre-stitch prevents scarring in reconstructions, and complex closures deploy battle-tested fibrinogen-thrombin sealants.
Malignant wounds, recalcitrant insurgents, are met with biologics infused with antimicrobial silver or metronidazole. New frontiers witness radiation burns giving way to stem cell foams beating cadaver grafts. Frostbite also employs cryopreserved cellular therapies that save tissue that was otherwise sentenced to necrosis.
In the case of battlefield blast injuries, hybrid biomatrices with clotting agents control hemorrhage while stimulation for regeneration is initiated where conventional packing descends into infection. Biologics don’t merely heal wounds—they recite survival narratives, rewriting last-ditch stands as comebacks once considered impossible.
Hospitals are still the war rooms for biologic deployment—ICUs amass grafts for burn patients, surgeons use amniotic membranes in amputations. Biologics here are heavy artillery: high-cost, high-risk interventions tracked around the clock.
Specialty clinics are guerrilla healers; wound centers in community settings use biologics like precision rifles a focused growth factor injection into a diabetic heel ulcer, a collagen matrix stuffed into a veteran’s pressure sore, skipping hospital bureaucracy for rapid, targeted healing.
The real revolution simmers in homes. Biologics now come through UPS coolers: grandparents administer nerve-growth gel to themselves under the supervision of telehealth nurses, construction workers spray placental powder onto on-site burns, weekend athletes apply growth-factor serums to recalcitrant blisters.
Telemedicine turns living rooms and kitchens into exam rooms; smartphone apps take pictures of wounds while AI algorithms adjust treatment regimens remotely. As biologics shrink from operating room fixtures to palm-sized vials and convenient sprays, healing essentially breaks free from institutional limitations.
Home health nurses increasingly provide and manage sophisticated cellular dressings, while community paramedics administer biologic first-response to complicated wounds. This democratization of front-line care serves patients exactly where life happens imperfectly, empowering individuals and depolarizing restoration.
North America holds the leading world position in sophisticated wound biologics. This leadership is a result of complex healthcare systems, prevalence of widespread chronic diseases such as diabetes, and facilitative reimbursement policies, especially U.S. Medicare coverage.
The United States leads innovation through regular FDA approval of next-generation biologic therapies, propelling adoption and establishing clinical benchmarks that shape world markets. High patient awareness and tremendous R&D investment further establish this region’s central place in both product development and use of advanced wound care products.
Europe holds a dominant market share of the wound biologics market, dominated by leading countries such as Germany and the UK. Strict regulatory environments in the EU focus on product safety and established efficacy, guaranteeing good quality standards.
A growing elderly populace contributes substantially to demand for complex wound management solutions, such as cellular and tissue-based products. Established healthcare systems and increased approval of advanced therapies underpin steady regional growth, as well as differences in national reimbursement systems affecting the speed of access.
The Asia-Pacific marketplace for wound biologics is the most rapidly growing, driven by giant populations in China and India with diabetes epidemics. Government and private healthcare spend, increasing hospital capacity, and growing medical tourism fuel adoption.
Cost-effective local manufacturing centers and the ability to improve product affordability and availability serve the region. Increasing clinician awareness and improving economic conditions provide significant opportunity for enhanced utilization of advanced biologic therapy within a wide range of health care settings over the next several years.
Latin America exhibits growth, especially in Mexico and Brazil, fueled by enhanced hospital infrastructure and diabetes-related government initiatives, though economic issues remain. The Middle East, specifically GCC nations such as the UAE, exhibits robust demand owing to high burn injury rates and high-value healthcare investments.
Africa has the highest access barriers mainly due to financial constraints; non-governmental organizations (NGOs) contribute significantly towards filling the gap through subsidized biologic programs and capacity development.
The global Wound Care Biologics market was valued at USD 1.8 billion in 2024.
The market is projected to grow at a CAGR of 7.7 % from 2025 to 2033.
Chronic wounds hold the largest market share.
The Asia-Pacific region is expected to witness the highest growth rate.
Major players include Smith & Nephew, Organogenesis Holdings Inc., MiMedx Group, Inc., Integra LifeSciences, Mölnlycke Health Care, Solsys Medical LLC, Anika Therapeutics, Inc., ACell, Inc.
1.1 Summary
1.2 Research methodology
2.1 Research Objectives
2.2 Market Definition
2.3 Limitations & Assumptions
2.4 Market Scope & Segmentation
2.5 Currency & Pricing Considered
3.1 Drivers
3.2 Geopolitical Impact
3.3 Human Factors
3.4 Technology Factors
4.1 Porters Five Forces Analysis
4.2 Value Chain Analysis
4.3 Average Pricing Analysis
4.4 M & A, Agreements & Collaboration Analysis
5.1 Wound Care Biologics Market, By Technology
5.1.1 Introduction
5.1.2 Market Size & Forecast
5.2 Wound Care Biologics Market, By Application
5.3 Wound Care Biologics Market, By End User
6.1 North America Wound Care Biologics Market, By Country
6.1.1 Wound Care Biologics Market, By Technology
6.1.2 Wound Care Biologics Market, By Application
6.1.3 Wound Care Biologics Market, By End User
6.2 U.S.
6.2.1 Wound Care Biologics Market, By Technology
6.2.2 Wound Care Biologics Market, By Application
6.2.3 Wound Care Biologics Market, By End User
6.3 Canada
7.1 U.K.
7.2 Germany
7.3 France
7.4 Spain
7.5 Italy
7.6 Russia
7.7 Nordic
7.8 Benelux
7.9 The Rest of Europe
8.1 China
8.2 South Korea
8.3 Japan
8.4 India
8.5 Australia
8.6 Taiwan
8.7 South East Asia
8.8 The Rest of Asia-Pacific
9.1 UAE
9.2 Turkey
9.3 Saudi Arabia
9.4 South Africa
9.5 Egypt
9.6 Nigeria
9.7 Rest of MEA
10.1 Brazil
10.2 Mexico
10.3 Argentina
10.4 Chile
10.5 Colombia
10.6 Rest of Latin America
11.1 Global Market Share (%) By Players
11.2 Market Ranking By Revenue for Players
11.3 Competitive Dashboard
11.4 Product Mapping